Tuberculosis is an infectious disease caused by the pathogen “Mycobacterium tuberculosis” – a bacterium with a waxy coating on its cellular surface due to the presence of mycolic acid.
Although tuberculosis is often successfully treated with antibiotics and sometimes prevented with the BCG vaccine – there are many cases of TB that: (A) are not prevented by the BCG vaccine (~80%) and/or (B) do not respond to antitubercular antibiotics.
As a result, tuberculosis kills ~1.5M people each year – and disability due to: TB-related lung damage and/or iatrogenic effects of antitubercular antibiotics. For these reasons, it is necessary to develop new treatments for TB.
New tuberculosis treatments
New tuberculosis treatments are desperately needed for a variety of reasons including:
- Suboptimal efficacy of prophylactic vaccines
- Ineffective antibiotic regimens
- Inefficient/long-term antibiotic regimens
- Adverse/iatrogenic effects from antibiotics
Latest TB drugs (Antibiotics)
A total of 3 antibiotics have been approved in the 2010s for the treatment of tuberculosis.

Prior to these antibiotics it had been decades since new antitubercular agents were developed and brought to market.
Pretomanid (PA-824)
- Class: Nitroimidazole
- Mechanism: Inhibits cell wall synthesis
- Approval date: 2019
Pretomanid (brand: Dovprela) is a nitroimidazole antibiotic used for the treatment of extensively drug-resistant tuberculosis (XDR-TB) or unresponsive multi-drug-resistant tuberculosis (MDR-TB).
It was first identified as in 2000 by PathoGenesis (Novartis subsidiary) after synthesizing and evaluating 100 nitroimidazopyran derivatives for antitubercular activity.
Pretomanid was found to exhibit bactericidal activity against static MTB isolates that survive in anaerobic conditions – and its effect was comparable to the existing antimicrobial agent metronidazole.
Interestingly, pretomanid was not the most potent antitubercular agent against MTB cultures – but it was the most active medication in MTB-infected mice after oral administration.
Pretomanid exerts a bactericidal effect against MTB by inhibiting mycobacterial cell wall synthesis via blocking of hydroxymycolate (mycolic acid) oxidation to ketomycolate. (R)
Usage of pretomanid is restricted due to the fact that medical professionals want to minimize antimicrobial resistance to its effect (if it’s frequently administered – occurrence of pretomanid resistance will increase).
Common side effects of pretomanid include: nerve damage/neuropathy; acne; vomiting; hypoglycemia; diarrhea; and liver inflammation.
Cost of pretomanid in the U.S. as of 2022 is ~$450-550 for a 26-tablet supply. Assuming at least 6 months of treatment – this will cost ~$3000-4000.
Delamanid
- Class: Nitroimidazole
- Mechanism: Inhibits cell wall synthesis
- Approval date: 2014
Delamanid (brand: Deltyba) is an antibiotic approved for the treatment of active multi-drug-resistant tuberculosis (MDR-TB).
Interestingly delamanid was approved for use in Europe, Japan, and South Korea in 2014 – but is not approved for use in the United States.
Preclinical and clinical studies showed that delamanid exhibited high potency, low risk of drug-drug interactions, and favorable tolerability. (R)
Delamanid is a prodrug (activated by a nitroreductase in MTB) that exerts mycobactericidal activity via inhibiting mycobacterial cell wall synthesis.
Specifically, it blocks methoxy-mycolic acid (MA) and keto-mycolic acid (MA) through the mycobacteria F420 system and generation of nitrous oxide. (R)
Common side effects of delamanid include: headache, dizziness, and nausea – and less frequently QT prolongation.
Pricing of delamanid is thought to be similar to bedaquiline and contingent upon country income status: (1) low-income: $900; (2) middle-income: $3,000; (3) high-income: $30,000.
Bedaquiline
- Class: Diarylquinoline
- Mechanism: Inhibits ATP synthase & bacterial respiration
- Approval date: 2012
Bedaquiline (brand: Sirturo) is a medication utilized for the treatment of MDR-TB (multi-drug-resistant tuberculosis).
It was discovered by a team led by Koen Andries at Janssen Pharmaceutica in 2004 – and its development cost an estimated: $455-747M (public sector) + $90-240M (Janssen Biotech) = ~$500-987M.
It received approval in 2012 by the U.S. FDA for tuberculosis treatment as part of a fast-track accelerated approval for specific cases of MDR-TB and XDR-TB.
The World Health Organization (WHO) and CDC (Centers for Disease Control) recommend that bedaquiline be reserved solely for individuals with MDR-TB wherein other recommended treatment regimens would be ineffective.
The reason bedaquiline usage is limited to drug-resistant TB infection is to reduce odds of bedaquiline-resistance via mutations in gene atpE and efflux pumps.
Bedaquiline is classified as a “diarylquinoline” antibiotic and exerts a bactericidal effect by blocking the proton-pumps for ATP synthase in mycobacteria.
The cost of bedaquiline for 6 months treatment varies depending on the country: (1) $900 low-income countries; (2) $3,000 middle-income countries; (3) $30,000 high-income countries.
Common side effects associated with bedaquiline include: nausea, joint & chest pain, and headache. It also carries a “black-box” warning for increased risk of death and arrhythmias (via QT prolongation).
There was concern associated with a trial of bedaquiline in which 10/79 bedaquiline recipients died – a figure much higher than 2/81 placebo recipients, however, when adjusting for: motor vehicle accidents (1 death); tuberculosis progression (5 deaths); deaths long after bedaquiline cessation (3 deaths) – this concern was unfounded. (R)
———————————————————-
Note: No “new” TB drugs are currently beyond Phase 2 clinical trials. There are Phase 3 clinical trials invovling various new combinations of drugs and/or already-approved antibiotics (e.g. clofazimine).

Telacebec (Q203)
- Class: Imidazopyridine amide (IPA)
- Mechanism: Bacterial respiration (cytochrome bc1 complex)
- Developer: Qurient Co., Ltd.
Telacebec (Q203) is an antibiotic of the imidazopyridine amide (IPA) class under investigation by Qurient Co., Ltd. for the treatment of tuberculosis.
It functions by interfering with the bacterial respiration of MTB – and specifically targets cytochrome bc1 complex to achieve this effect.
A report in 2013 reported the discovery of “Q203” – describing it as a potent clinical candidate for the treatment of tuberculosis. (R)
Q203 inhibited the growth of MDR and XDR tuberculosis isolates in culture broth medium in the low nanomolar range and was effective in a mouse-model of TB at a dose less than 1 mg/kg bodyweight – which showcases its potency of action.
An open-label, proof-of-concept trial (2020) involving 61 patients with tuberculosis revealed that increasing doses of telacebec was associated with greater reductions in MTB sputum load – and there were no adverse drug reactions. (R)
A report (2021) noted that telacebec is highly lipophilic which could be a reason as to why it appears highly-effective against tuberculosis – while maintaining favorable pharmacodynamic, pharmacokinetic, and toxicological properties. (R)
A study (2022) showed that telacebec was well-tolerated and safe in 56 healthy adults – such that it caused no adverse reactions. (R)
Note: Verapamil and small-molecule inhibition of cytochrome bd appear to enhance the efficacy of telacebec. (R1, R2)
BTZ-043
- Class: Benzothiazinone
- Mechanism: Inhibits cell wall synthesis (DprE1)
- Developer: University of Munich; German Center for Infection Research (DZIF); EDCTP; Radbound University
BTZ-043 is a benzothiazinone antibiotic under investigation by numerous agencies (University of Munich, German Center for Infection Research, EDCTP, and Radbound University) for the treatment of tuberculosis.
BTZ-043 functions as an efficient inhibitor of mycobacterial cell wall synthesis via blocking DprE1 necessary for synthesis of D-arabinofuranose (a component of arabinogalactan & arabinomannan). (R)
In vitro MICs (minimum inhibitory concentrations) for BTZ-043 against MTB range from 1-30 ng/mL.
It appears as though BTZ-043 is active against all tested MTB isolates including MDR (multi-drug-resistant) and XDR (extensively-drug-resistant) strains.
In mouse models, BTZ-043 exhibits superior activity over isoniazid (INH) – which becomes most prominent after 2 months of administration.
Preliminary evidence suggests that BTZ-043 has low toxicological potential and is well-tolerated – and phototoxicity, genotoxicity, mutagenicity studies were all negative.
Delpazolid
- Class: Oxazolidinone
- Mechanism: Inhibits protein synthesis (23S ribosome)
- Developer: LegoChem Biosciences, Inc.
Delpazolid (LCB01-0371) is an oxazolidinone antibiotic under investigation by LegoChem Biosciences, Inc. for the treatment of tuberculosis.
It acts as an antitubercular agent by inhibiting mycobacterial protein synthesis by binding to a site on the bacterium’s 50S subunit 23S ribosomal RNA.
An in vitro susceptibility study (2018) indicated that delpazolid exhibited antibacterial activity comparable to linezolid – against strains of drug-resistant (MDR/XDR) tuberculosis. (R)
A report (2020) stated that delpazolid can improve the minimal bactericidal concentration of MTB (H37Rv) and significantly reduce resistance rates, especially of MDR-TB isolates, relative to linezolid. (R)
A study (2022) involving patients with TB noted that delpazolid (400 mg, b.i.d.) is effective against M. tuberculosis and is significantly less toxic than other oxazolidinones. (R)
GSK-656
- Class: Oxaborole
- Mechanism: Inhibits protein synthesis (LeuRS)
- Developer: GlaxoSmithKline (GSK)
GSK-656 is a novel oxaborole antibiotic under investigation by GlaxoSmithKline (GSK) for the treatment of tuberculosis.
It was discovered in 2017 after synthesizing and evaluating a novel series of 3-aminomethyl 4-halogen benzoxaboroles as M. tuberculosis leucyl-tRNA synthetase (LeuRS) inhibitors. (R)
It exerts an antimycobacterial effect via inhibiting protein synthesis of M. tuberculosis bacteria – specifically by blocking the enzyme leucyl-tRNA synthetase (LeuRS).
Some research suggests that GSK-656 may be able to replace an oxazolidinone without mitochondrial protein synthesis (MPS)-associated toxicity. (R)
A study (2020) suggests that GSK-656 exhibits potent efficacy against M. tuberculosis as evidenced by MICs less than 0.5 mg/L for 102 of 105 MTB isolates; MIC50 = 0.063 mg/L & MIC90 = 0.25 mg/L. (R)
Preliminary evidence (2019) suggests that GSK-656 is safe and generally well-tolerated in humans after single and multiple doses. (R)
OPC-167832
- Class: Carbostyril derivative
- Mechanism: Inhibits cell wall synthesis (DprE1)
- Developer: Otsuka Pharmaceutical Inc.
OPC-167832 is a carbostyril derivative under investigation by Otsuka Pharmaceutical Inc. for the treatment of tuberculosis.
After screening a library of carbostyrils, researchers optimized 3,4-dihydrocarbostyril derivatives and identified OPC-167832 as exhibiting strong antitubercular activity.
OPC-167832 exerts an antimycobacterial effect via inhibition of DprE1 enzymes of mycobacteria – which are needed for synthesis of D-arabinofuranose (a component of arabinogalactan & arabinomannan) to form cell walls.
In vitro investigations indicate that OPC-167832 exhibits very low MICs against lab strains of MTB (H37Rv) and Kurono strains with resistance to various antitubercular agents (rifampicin, isoniazid, ethambutol, streptomycin, pyrazinamide); MIC values were 10 to 1000 times lower than other anti-TB drugs. (R)
Interestingly, OPC-167832 shows minimal or no activity against standard nonmycobacterial aerobic/anaerobic bacteria – suggesting little effect on bacterial flora of the host.
In a mouse model of chronic TB, OPC-167832 facilitates a potent bactericidal effect at a dose of 0.625 mg/kg bodyweight and exhibits substantial synergistic effects in 2-drug combinations with delamanid, bedaquiline, or levofloxacin.
Fobrepodacin (SPR720)
- Class: Benzimidazole
- Mechanism: Inhibits DNA synthesis
- Developer: Spero Therapeutics & GMRI
SPR720 is a novel benzimidazole antibiotic under investigation by Spero Therapeutics & GMRI for the treatment of tuberculosis.
SPR720 is a phosphate prodrug of SPR719 which exerts an antimycobacterial effect by inhibiting the ATPase subunits of DNA gyrase (GyrB) of mycobacteria – and topoisomerase when present. (R)
Phase 1 clinical trials indicate that once-daily dosing of SPR720 is safe and generally well-tolerated with side effects limited to gastrointestinal complains and headache. (R)
SQ109
- Class: 1,2-ethylene diamine (Ethambutol analogue)
- Mechanism: Inhibits cell wall synthesis (MmpL3)
- Developer: Sequella
SQ109 is a 1,2-ethylene diamine-based ethambutol analogue antibiotic that is under investigation by Sequella for the treatment of tuberculosis.
SQ109 was first described in 2005 as displaying promising in vitro and in vivo antitubercular activity – with favorable targeted tissue distribution profiles. (R)
A study (2007) in mice found that combining SQ109 with Isoniazid, Rifampicin, and Pyrazinamide provided a new and very effective treatment regimen for TB with faster kill times than standard regimens. (R)
It exerts an antimycobacterial effect by interfering with a mycolic acid transporter called “MmpL3” which is needed by MTB to incorporate mycolic acid into its cell wall. (R)
SQ019 exhibits a delayed-onset of action in animal models of tuberculosis – requiring 8-to-12 weeks of treatment to achieve maximal efficacy – and is believed to exhibit similar binding and distribution to bedaquiline and clofazimine. (R)
Sutezolid
- Class: Oxazolidinone
- Mechanism: Inhibits protein synthesis (23S ribosome)
- Developer: Sequella & TB Alliance
Sutezolid is an oxazolidinone antibiotic under co-investigation by Sequella and TB Alliance for the treatment of tuberculosis.
It exerts an antimycobacterial effect via interacting with the 50S subunit 23S ribosomal DNA of mycobacteria – which inhibits mycobacterial protein synthesis.
A study (2014) found that sutezolid facilitated significant mycobactericidal activity in sputum and blood of persons with pulmonary tuberculosis – and that it was generally safe and well-tolerated. (R)
A single ascending-dose study (2022) revealed that sutezolid was well-tolerated when administered at 300 mg, 600 mg, 1200 mg, and 1800 mg in healthy adult subjects while fasted. (R)
Sudapyridine (WX-081)
- Class: Diarylquinoline
- Mechanism: Inhibits ATP synthase & bacterial respiration
- Developer: Shanghai Jiatan Biotech
Sudapyridine is a novel analogue of bedaquiline under investigation by Shanghai Jiatan Biotech for the treatment of tuberculosis.
It exerts an antimycobacterial effect by targeting and inhibiting F-ATP synthase activity of mycobacteria which blocks mycobacterial respiration.
Sudapyridine exhibits favorable in vitro activity against clinically isolated strains of M. tuberculosis, including both drug-sensitive (DS) and drug-resistant (DR) strains.
The in vivo effectiveness of sudapyridine in mice is dose-dependent and analogous to that of bedaquiline – an approved medication to treat tuberculosis. (R)
Though the efficacy of sudapyridine appears similar to bedaquiline, it is associated with lower risk of cardiotoxicity than bedaquiline. (R)
Overall, it seems as though sudapyridine is extremely similar to bedaquiline in efficacy (which makes sense given that it’s a BDQ analogue) with a potentially improved safety profile regarding cardiotoxicity.
TBA-7371
- Class: Azaindole
- Mechanism: Inhibits cell wall synthesis (DprE1)
- Developer: Astra Zeneca; TB Alliance; GMRI
TBA-7371 is an azaindole antibiotic compound that was developed collaboratively by Astra Zeneca, TB Alliance, and the GMRI for the treatment of tuberculosis.
It is a chemical derivative of 1,4-azaindoles and was synthesized via scaffold morphing of an imidazopyridine compound.
TBA-7371 facilitates an antimycobacterial effect by inhibiting DprE1 enzymes of mycobacteria thereby blocking the synthesis of D-arabinofuranose (a component of arabinogalactan & arabinomannan) and subsequent cell wall formation.
Preliminary research suggests TBA-7371 exhibits a favorable in vitro safety profile without cytotoxicity up to 100 uM and good efficacy in mouse models. (R)

Pyrifazimine (TBI-166)
- Class: Riminophenazine
- Mechanism: Inhibits ion transport & bacterial respiration
- Developer: GATDD, IMM, CAMS, PUMC
Pyrifazimine (TBI-166) is an orally-bioavailable clofazimine analogue of the riminophenazine class that is under investigation by various organizations (GATDD, IMM, CAMS, PUMC) for the treatment of tuberculosis.
Clofazimine is a drug that has been used since 1962 to treat leprosy (caused by mycobacteria) and has been incorporated into short-course MDR-TB regimens.
However, because clofazimine has suboptimal pharmacokinetics & pharmacodynamics (PK/PD) – it accumulates in the tissue and causes tanning/reddish discoloration and takes months to eliminate.
Pyrifazimine was engineered to have improved PK/PD properties (relative to clofazimine) while facilitating a potent antitubercular effect with less skin discoloration.
It exerts an antimycobacterial effect by inhibiting both ion transport and bacterial respiration. (R)
Research (2022) involving in vitro and in vivo experiments in mice indicates that pyrifazimine exhibits synergistic antitubercular activity when combined with other antitubercular agents. (R)
TBAJ-876
- Class: Diarylquinoline
- Mechanism: Inhibits ATP synthase & bacterial respiration
- Developer: TB Alliance & University of Auckland
TBAJ-876 is a novel antitubercular compound of the diarylquinoline classification under co-investigation by the TB Alliance & University of Auckland for the treatment of tuberculosis.
TBAJ-876 functions by interfering with energy production for M. tuberculosis via direct inhibition of ATP synthase and bacterial respiration.
In animal models, TBAJ-876 appears more effective and potent against TB than bedaquiline and its predecessor TBAJ-587 (with a lower predicted clinical dose than either compound) – and also safer. (R)
It seems as though this is a good candidate to improve upon/replace bedaquiline in current TB regimens.
TBAJ-587
- Class: Diarylquinoline
- Mechanism: Inhibits ATP synthase & bacterial respiration
- Developer: TB Alliance & University of Auckland
TBAJ-587 is a diarylquinoline antibiotic under investigation by TB Alliance and University of Auckland for the treatment of tuberculosis.
It exerts an antimycobacterial effect via inhibiting F-ATP synthase of M. tuberculosis – thus interfering with bacterial respiration and energy production. (R)
In mouse models, TBAJ-587 is more effective against wild-type and isogenic mutant tuberculosis strains than bedaquiline whether administered alone or in conjunction with other antitubercular compounds. (R)
Researchers have found that TBAJ-587 appears to reduce the emergence of resistance to diarylquinolines and pretomanid.
Like TBAJ-876 – the compound TGAJ-587 exhibits superior potency, lower lipophilicity, and superior cardiovascular risk profiles than other diarylquinolines. (R)
TBI-223
- Class: Oxazolidinone
- Mechanism: Inhibits protein synthesis (23S ribosome)
- Developer: TB Alliance & IMM
TBI-223 is an oxazolidinone antibiotic under investigation by TB Alliance & IMM (Institute of Materia Medica) for the treatment of tuberculosis.
Like other oxazolidinones, TBI-223 exerts antimycobacterial actions via interaction with the 50S subunit 23S of mycobacteria DNA – thus blocking mycobacterial protein synthesis.
It appears to have low mitochondrial protein synthesis (MPS) activity and an improved safety profile versus linezolid.
Macozinone (PBTZ-169)
- Class: Benzothiazinone
- Mechanism: Inhibits cell wall synthesis (DprE1)
- Developers: iM4TB + Bill & Melinda Gates Foundation
Macozinone is a benzothiazinone antibiotic under investigation by iM4TB and Bill & Melinda Gates Foundation for the treatment of tuberculosis.
It is a piperazinobenzothiazinone derivative optimized by medical chemistry from the lead compound BTZ-043.
Macozinone is chemically advantageous over BTZ-043 in that it is easier to synthesize and exhibits superior pharmacodynamics.
It exerts an antimycobacterial effect by inhibiting mycobacterial cell wall synthesis via targeting DprE1 enzymes of mycobacteria.
Preclinical research suggests that macozinone is synergistic with bedaquiline, clofazimine, delamanid, and sutezolid in its antimycobacterial action. (R)
Phase 1 clinical trials indicate that macozinone appears safe and tolerable in humans. (R)
GSK-286
- Class: N/A
- Mechanism: Inhibits cholesterol catabolism
- Developers: GlaxoSmithKline (GSK)
GSK-286 is an antibiotic under investigation by GlaxoSmithKline (GSK) in collaboration with TB Drug Accelerator & GMRI – for the treatment of tuberculosis.
Its exact mechanism of action remains unclear but it is believed to target and inhibit mycobacterial cholesterol catabolism.
GSK-286 is effective against multi-drug-resistant (MDR), extensively-drug-resistant (XDR), and drug-sensitive (DS) tuberculosis in human macrophages and culture mediums – with no cross-resistance to known antitubercular agents. (R)
GSK-286 has shown efficacy in various mouse models of tuberculosis and appears safe.
BVL-GSK098
- Class: Amido piperidine
- Mechanism: Inhibits transcriptional regulators
- Developers: BioVersys AG & GSK
BVL-GSK098 is a compound that was discovered by BioVersys’ “TRIC” (Transcriptional Regulator Inhibitory Compound) platform in collaboration with GSK and the Pasteur Institute of Lille – for the treatment of tuberculosis.
It is intended to be utilized as part of a combination treatment protocol with ethionamide (Eto)/prothionamide (Pto) in the treatment of tuberculosis.
Although ethionamide and prothionamide are useful in the treatment of TB, they have suboptimal bio-activation within MTB – and thus high doses are needed to achieve clinical efficacy which causes increased rates of adverse events (GI intolerance & hepatotoxicity). (R)
BVL-GSK098 functions by acting on bacterial transcriptional regulators – stimulating bio-activation pathways for ethionamide and prothionamide to increase their efficacies and reduce likelihood of ethionamide/prothionamide resistance.
Animal studies indicate that BVL-GSK098 could allow for a dosage-reduction of ethionamide/prothionamide by at least 3-fold – which should reduce rates of adverse events and simultaneously enhance antitubercular treatment efficacy.
Pre-Clinical (GLP)
These are compounds that are in pre-clinical studies with “good laboratory practices” (GLP).
Sanfetrinem: An orally-available tricyclic beta-lactam (trinem) antibiotic that exhibits antimycobacterial action via targeting cell wall biosynthesis. Sanfetrinem cilexetil (prodrug) is also under investigation. (R)
FNDR-20081: A promising first-in-class compound that exerts a multi-targeted effect to kill mycobacterium tuberculosis. (R)
OTB-658: An oxazolidinone antibiotic that exerts an antimycobacterial effect by inhibiting mycobacterial protein synthesis. Preliminary evidence suggests that it inhibits MTB on par with linezolid. (R)
FNDR-20364: An inhalable antimicrobial peptide with strong in vivo & in vitro activity in animal models of tuberculosis infection. It kills TB by inhibiting ribosome-associated GTPase activity blocking protein translation.
TB47: A telacebec analogue that targets the QcrB in the electron transport chain of mycobacteria. It has shown synergistic activity with clofazimine and linezolid. (R)
GSK839: Identified as a possible candidate for tuberculosis treatment via a sponsored program. It is a “tetrazole benzene sulfonamide” antibiotic that inhibits TrpAB (tryptophan synthase) of M. tuberculosis in a selective manner.
MBX-4888A: A spectinamide antibiotic (semisynthetic spectinomycin analog) that exhibits selective ribosomal inhibition and narrow-spectrum antitubercular action. It appears safe and effective in murine models of TB infection – and is synsergistic with other antitubercular agents.
Targets of antitubercular agents
Below are various pharmacological targets of antitubercular agents.
Cell Wall Synthesis
Peptidoglycan Layer
- Mur ligases
- D-alanine:D-alanine Ligase (DDL)
- translocase 1 (MurX or Mra Y)
- Lipid II
- D,D-transpeptidase
- L,D-transpeptidase
Arabinogalactan Layer
- DPRE1 Noncovalent Inhibitors
- Arabinosyl transferase
- WecA inhibitor
- DprE1 Covalent Inhibitors
Mycolic Acid Layer
- MmpL3 Inhibitor
- InhA Inhibitor
- KasA Inhibitors
- Unknown Target Mycolic Acid Inhibitors
DNA Replication
- DNA gyrase
DNA Transcription
- RNA Polymerase
RNA Translation
- rRNA / Ribosome
Energy Metabolism
- ATP-Synthase
- Cytochrome bc1-aa3
- NADH Dehydrogenase Type II
- MenG Inhibitor
Proteolysis & Proteostasis
- ClpC
- ClpP1P2
Cellular Metabolism
- Unknown Target Cholesterol Metabolism
- Tryptophan Synthase (TrpAB)
- Aspartate decarboxylase (PanD)
- Efflux transport of antibiotics (efpA)
New tuberculosis treatment regimens
Permutations of tuberculosis antibiotics are being evaluated to determine whether new regimens can improve upon preexisting: (1) efficacy (cure rates); (2) tolerability, side effects, safety; and/or (3) minimal required duration of treatment.
-
Bedaquiline + Clofazimine + Rifapentine + Levofloxacin + Linezolid
-
Bedaquiline + Delamanid + Linezolid + Levofloxacin + Clofazimine
-
Bedaquiline + Delamanid
-
Bedaquiline + Linezolid + Levofloxacin + Pyrazinamide + Ethionamide or Isoniazid or Terizidone
-
Bedaquiline + Pretomanid + Linezolid
-
Bedaquiline + Pretomanid + Linezolid + Moxifloxacin
-
Bedaquiline + Moxifloxacin + Linezolid
-
CC-11050 + Auranofin + Everolimus + Vitamin D3 + Rifabutin-modified TB therapy
-
Delamanid + OBR (optimized background regimen)
-
Delamanid + Bedaquiline + OPC-167832
-
Delpazolid + Bedaquiline + Delamanid + Moxifloxacin
-
Isoniazid + Rifampicin + Pyrazinamide + Moxifloxacin + Ethambutol
-
Pravastatin + Rifafour + Vitamin B6
-
Pretomanid + Moxifloxacin + Pyrazinamide
-
Sutezolid + Bedaquiline + Delamanid + Moxifloxacin
A primary reason new treatment regimens are under investigation is to determine whether it’s possible to shorten TB treatment duration.
Drug-sensitive TB
- One study found that 4-month treatment regimens with rifapentine (RFP) and moxifloxacin (MFX) were non-inferior to standard 6-month treatment regimens without these agents – with no increase in adverse events. (R)
- SimpliciTB trial: Evaluating Bedaquiline + Pretomanid + Moxifloxacin + Pyrazinamide – to determine whether it’s effective as a 4-month regimen. (Some have noted that this is unlikely to be used regularly on the basis that medications need to be preserved to prevent antibiotic resistance.)
- TRUNCATE-TB trial: Evaluating combinations of currently-approved antitubercular agents (rifampicin, isoniazid, pyrazinamide, ethambutol, linezolid, clofazimine, rifapentine, levofloxacin, bedaquiline) – given daily 8-weeks (2-month regimen).
- RIFASHORT & ReDEFINe studies: Evaluating the risk-benefit ratio of higher doses of rifampicin (RIF) in drug-sensitive TB cases – on the basis that it may accelerate mycobacterial sputum clearance without increase in toxicity.
Drug-resistant (DR) TB
- BEAT-TB: Assessing the efficacy of BDQ, LZD, DLM, LFX, and CFZ vs. standard-of-care – over a 6-month period.
- STREAM2: Evaluating a shorter regimen for RR-TB and MDR-TB in a multi-armed study with 4 regimens based upon WHO practice; Bangladesh regimen; 40-week all oral regimen; and a 28-week oral regimen after an 8-week intense regimen with INH & Kanamycin.
- NEXT trial: Compared 6-9 months of LZD, BDQ, LFX, PZA, ETH or INH to standard-of-care.
- DELIBERATE: Evaluating the safety and pharmacokinetics of BDQ + DLM.
- endTB: Evaluating various combinations of an all-oral regimen for 39 weeks.
- ZeNix trial: Reviewing treatment regimens for patients with pre-XDR or XDR-TB. Using BDQ, Pa, and either LZD (BPaL) or a placebo for a duration of 26 weeks. Aim is to evaluate rates of sputum conversion.
Adjunct treatments for TB
- Immunomodulation: A balanced immune response appears to increase survival in TB.
- Inflammation optimization: Deficient and excessive inflammation are both associated with TB disease morbidity and mortality.
- Host-directed therapy: Host-directed therapies tailored to patient LTA4H genotypes may counteract detrimental effects of suboptimally high/low inflammation. (R)
- Vitamin D: Low levels of vitamin D may impair immune responses and increase TB survival.
- Everolimus: A targeted cancer drug that blocks cancer growth.
- Auranofin: An anti-inflammatory agent used to treat rheumatoid arthritis.
- CC-11050: A novel anti-inflammatory compound.
What about new tuberculosis vaccines?
There are many types of vaccines in development for tuberculosis – and several promising candidates are in Phase 3 clinical trials.
Read: New tuberculosis vaccines.
New modes of antibiotic delivery for TB
Inhalable medications: To more effectively target the lungs without as many significant systemic effects (which should reduce side effect burden).
- Example: Arikayce (inhalable amikacin) – an aminoglycoside antibiotic that has been used for over 40 years to treat MDR-TB.
Targeted drug delivery: A system of specifying the drug moiety directly into its targeted area (organ, cellular, subcellular level of specific tissue) to overcome the non-specific toxic effect of conventional drug delivery. (R)
- To achieve this a “magic bullet” concept was developed and led to scientists environing nanometer-sized devices (nanomedicine).
- Various carrier systems such as colloidal (vesicular & multi-particulate) carriers, polymers, and cellular/subcellular systems are being tested/investigated.
Obviously if we could design a drug that could be mainlined directly into M. tuberculosis within the lungs – without affecting other areas of one’s body – we could cure MTB infections rapidly and patients would experience no significant side effects/adverse events.
A major problem now is that M. tuberculosis has a waxy-outer coating that buffers/protects it from antibiotic penetration – hence the need for combination therapy – and high doses.
- Certain antibiotics like ethambutol in TB treatment regimens function primarily by targeting the outer waxy-coating of MTB so that other antibiotics can penetrate the bacteria and kill it more effectively.
- High doses of antibiotics are required because antibiotics do not concentrate solely within MTB-infected regions – as a result only a small amount of the dose actually makes it to the necessary destination (MTB infection).
- If we could concentrate specific antibiotics specifically within MTB or within MTB-infected regions (i.e. the lungs) – cure rates would be higher and time-to-cure would be shorter.
General tuberculosis information
Below are some general statistics about tuberculosis infection (in the event that you’re not familiar with TB).
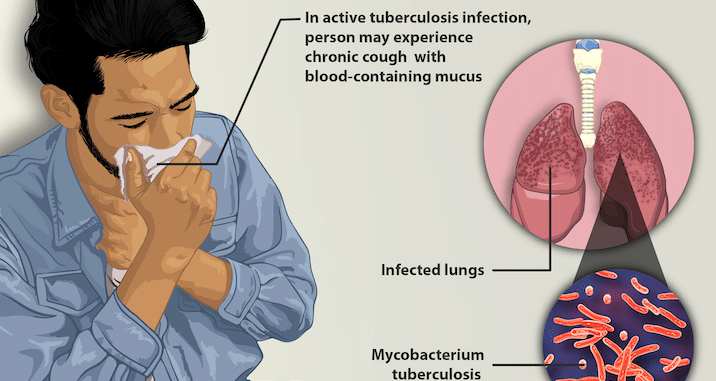
Image credit: MyUpchar
Location & rates of tuberculosis infection
Rates of tuberculosis infection differ significantly by location. (R)
- <25 cases per 100K people: U.S., Canada, Western Europe, Australia, Japan.
- ~26 cases per 100K people: China, Eastern Europe, Central/South America, northern Africa.
- 100 cases per 100K people: India, sub-Saharan Africa, Micronesia, Southeast Asia.
Status of tuberculosis infections
Latent TB: Mycobacterium tuberculosis bacteria are in your body (usually lungs) but your immune system keeps them in check such that they don’t spread or inflict significant damage and you’re not contagious.
- Over 80% of TB cases are associated with longstanding, untreated latent TB infections.
- Up to 13 million people in the U.S. have latent TB infection – many of whom traveled outside of the U.S. and were infected in areas where TB is endemic.
- 1/3 of the global population (~2.3 billion individuals) is likely infected with latent TB at this moment. (R)
Active TB: Mycobacterium tuberculosis bacteria are in your body (usually lungs) and are spreading/inflicting damage such that it’s damaging your body and is contagious to others.
- According to the World Health Organization (WHO), around 8 million people develop active tuberculosis each year globally.
- 1/10 people infected with tuberculosis (~10%) develop “active” infection wherein it becomes contagious and inflicts damage upon the body. (R)
- If untreated or unresponsive to treatment, active tuberculosis can damage one’s lungs and lead to a slow, painful death.
Types of tuberculosis (R)
- Drug-sensitive TB: Responds well to conventional anti-tuberculosis antibiotic therapy – usually over a period of 6-9 months.
- Drug-resistant (DR): Resistance to at least one first-line anti-TB drug (e.g. rifampin, pyrazinamide, isoniazid, ethambutol, etc.).
- Multi drug resistant (MDR): Resistant to multiple first-line anti-TB drugs (e.g. rifampin, pyrazinamide, isoniazid, ethambutol, etc.).
- Extensively drug resistant (XDR): Resistant to first-line and second-line TB drugs like MDR-TB but with additional resistance to fluoroquinolones and bedaquiline.
Note: Some use classifications of TB based on the specific drug(s) to which it is resistant (e.g. rifampin-resistant TB).
How many people are infected each year with tuberculosis?
It’s estimated that there are 10.4 million new cases of tuberculosis each year. (R)
It’s also estimated that 1/3 of new cases (~3 million) remain unknown to health systems – and many end up not receiving adequate treatment.
How many people die each year of tuberculosis?
~1.8 million people die each year as a result of tuberculosis.

Biggest problems with current tuberculosis treatments & prophylactics…
Antibiotics
- Efficacy: Not always effective – particularly in cases of drug-resistant, multi-drug resistant, and extensively drug resistant. As a result of suboptimal efficacy, people die.
- Adverse events: Hearing loss, tinnitus, blindness (ethambutol), vestibulotoxicity (balance issues), neuropathy, C. diff, etc.
- Long-term: TB generally requires antibiotic treatment for 6-9 months (assuming the specific strain is responding to treatment).
Side effects of tuberculosis antibiotics (R)
Although antimycobacterial agents (i.e. anti-TB drugs) are sometimes well-tolerated without significant side effects, if certain side effects occur they can have a lasting negative impact on one’s health, wellbeing, and/or overall functioning.
The most noteworthy side effects (in my opinion) include: blindness; hearing loss; tinnitus; liver damage; kidney damage; tendonitis; peripheral neuropathy; and QT prolongation. In many cases these are not fully reversible.
- Rifampicin (RIF): Hepatotoxicity; nausea; dyspepsia; abdominal pain; CYP450 interactions.
- Isoniazid (INH): Hepatotoxicity; peripheral neuropathy; optic neuritis; CYP450 interactions.
- Pyrazinamide (PZA): GI disturbances; hepatotoxicity; gout.
- Ethambutol (EMB): Optic neuropathy/optic neuritis. (Read more: Ethambutol & Ocular Toxicity).
- Levofloxacin (LFX): QT prolongation; tendonitis; hypoglycemia; psychiatric complications.
- Moxifloxacin (MFX): QT prolongation; tendonitis; hypoglycemia; psychiatric complications.
- Bedaquiline (BDQ): QT prolongation; CYP450 interactions.
- Linezolid (LZD): Myelosuppression; dysglycemia; peripheral neuropathy; optic neuritis.
- Clofazimine (CFZ): Hepatotoxicity; GI disturbance; neurological disturbance; QT prolongation; altered skin pigmentation (tanned appearance).
- Cycloserine (CYS): Psychiatric disturbance; peripheral neuropathy.
- Delamanid (DLM): QT prolongation; CYP450 interactions.
- Aminoglycosides (AMK, CAP, STR): Ototoxicity; nephrotoxicity.
- Ethionamide (ETH)/Prothionamide (PRO): Psychiatric complications; peripheral neuropathy; hepatotoxicity; hypothyroidism; dysglycemia.
- p-Aminosalicyclic acid (PAS): Hepatotoxicity; dysglycemia; hypothyroidism.
- Amoxicillin-Clavulanate with Meropenem or Imipenem-Cilastatin: Diarrhea; candidiasis.
Duration of TB treatment with antibiotics
Drug-sensitive tuberculosis is treated for ~6-9 months on average depending on the specific case – but this is still a long time to be on 2-4 antibiotics.
Drug-resistant tuberculosis is treated anywhere from 9-21 months on average depending upon the specific case (greater severity = longer necessary treatment duration).
Drug-sensitive tuberculosis regimen (6 months)
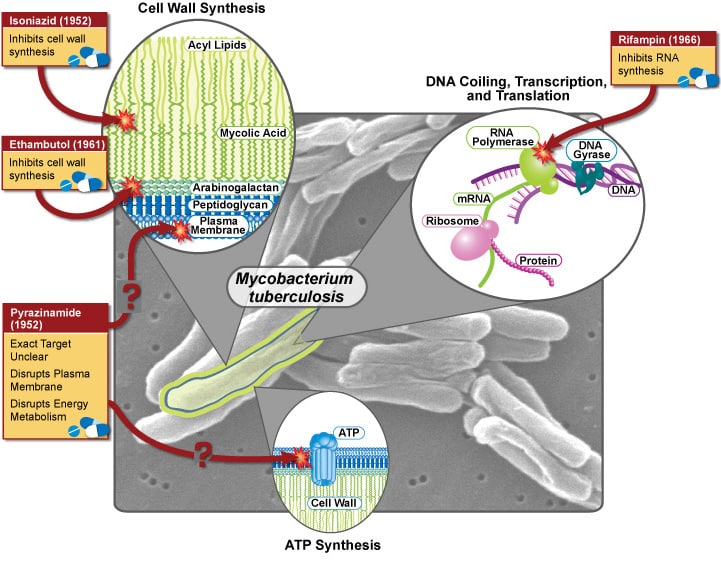
Resized image credit: NIAID
Intensive phase (2 months): Rifampicin; Isoniazid; Pyrazinamide; Ethambutol.
Continuation phase (4 months): Rifampicin + Isoniazid.
Note: The continuation phase is extended to 7 months if lung cavitation (i.e. cavitary lesion) is present; persistent sputum growth is exhibited; and/or PZA cannot be used due to mono-resistance or side effects.
Multidrug resistant TB (MDR-TB) regimen (9-21 months)
- First-line: Levofloxacin or Moxifloxacin with: Bedaquiline + Linezolid + Clofazimine + Cycloserine
- Second-line: Delamanid or Pyrazinamide or Ethambutol or Amikacin or Streptomycin
- Third-line: Ethionamide or Prothionamide or Imipenem-Cilastatin/Clavulanate or Meropenem/Clavulanate or p-Aminosalicylic acid or high-dose Isoniazid
Vaccines (BCG)
Presently the only vaccine that’s approved by health organizations to prevent tuberculosis infection/disease is called the BCG (Bacillus Calmet-Guerin) vaccine.
BCG vaccination involves subcutaneously administering a specific “weak” live strain of the bacterial species M. bovis (Mycobacterium bovis) called “BCG.”
Because M. bovis is closely related to M. tuberculosis – it is thought that training the immune system to overcome a “BCG” (a specific M. bovis strain) will yield cross-immunity to protect against M. tuberculosis infection/disease.
Suboptimal efficacy & impermanent effect
The effectiveness of BCG vaccination for the prevention of tuberculosis infection/disease is considered suboptimal.
An analysis of BCG by Okafor et al. (2022) stated that the effectiveness of BCG vaccination in healthcare workers is “not definite.” (R)
Brett & Severn (2020) found that BCG protects against drug-susceptible TB infection and death in immunocompetent persons – but evidence for this finding is from low-to-moderate quality and outdated (1930-1980s) studies. (R)
- This finding does not support the idea that BCG protects against TB infection/disease in every immunocompetent person who receives it.
- Instead, it suggests BCG provides a higher degree of protection in terms of infection rate and disease severity – on average, than no BCG vaccination.
- Researchers acknowledged that BCG’s efficacy against drug-resistant forms of TB infection/disease remains unknown.
Other researchers believe that BCG’s efficacy depends upon a variety of individual-specific variables including: (1) age at vaccination; (2) immune profile/expression; (3) genetics/epigenetics; and (4) environment.
Some evidence suggests that the BCG vaccine protects ~20% (1 out of 5) children from TB infection/disease – but this is still a low rate of protection (~80% unprotected even with BCG vaccination). (R)
Additionally, researchers believe that adults are far less protected from BCG vaccination than children – meaning significantly less than 20% of adults who receive the vaccine will derive significant protective benefit.
It is also thought that the protective effect derived from BCG vaccination against TB infection/disease may be limited in its duration – such that it fades significantly or back to near-zero protection years after receiving BCG (assuming no “booster” was administered).
My ideas for new TB treatments
Just an assorted brain-vomit of ideas that I’ve had (some of which might be undergoing research).
- New antibiotic compounds: Discovered via machine-learning AI and humans.
- Antibiotic combinations: Hollow fiber models should be used to develop new drug combinations that can be used to eradicate TB in a more efficient manner.
- Phages, viruses, bacteria, fungi, archaea, etc.: Anything that can compete with or destroy TB without damaging the body – this includes bacteriophages, viruses, other bacteria, fungi, archaea, etc.
- Targeted antibiotic therapy: Rather than systemic. Closest thing we’ve got right now is inhalable liposomes – but this is probably a good option to further explore.
- Antimicrobial polymers: Polymers with the ability to kill or inhibit the growth of mycobacteria.
- Body temperature manipulation: Such as whole body hyperthermia should be investigated as an adjunct intervention.
- RNAi therapeutics: Agents that enhance host antibacterial capacity against TB. (R)
- Surgery: Improve upon existing surgical techniques if possible. Explore outcomes in XDR-TB.
- Immunomodulation: Various immunomodulatory agents.
- Analogues & derivatives: Of existing TB drugs that may be equally as deadly towards MTB as parent compounds but less toxic to users (and thus higher-doses can be used and/or fewer side effects occur). Derivatives of various drugs like thioridazine should be investigated as well.
- Dietary supplements: Need to be further investigated and explored as adjuncts such as: high-dose vitamin C; high-dose melatonin; garlic extract; vitamin D3; high-dose NAC; etc.
- Gaseous nitric oxide: Appears to exhibit antimycobacterial action.
- Repurposing existing drugs: Thioridazine, verapamil, etc. appear to exhibit antimycobacterial action.
My ideas for new TB vaccines
- mRNA TB vaccine: Piggyback off of mRNA tech utilized for COVID-19 vaccines and develop a TB vaccine.
- IV BCG: Needs to be tested ASAP in humans. Found to be significantly more effective in monkeys than the standard intramuscular BCG vaccine.
- Post-infection vaccine: Not only should we have a vaccine to protect against TB infection, but we should work on developing a vaccine that prevents/minimizes odds of reinfection (post-infection). (Many vaccines that prevent infection do NOT prevent reinfection.
Challenges associated with TB drug development…
Developing new drugs for the treatment of tuberculosis is an endeavor that’s been abandoned by most profit-motivated pharmaceutical companies. (R)
For this reason, the development of new tuberculosis medications has been largely subsidized by various countries (i.e. governments); philanthropy; and academic institutions.
Funding: Financial investment in new antibiotics for the treatment of tuberculosis is extremely limited. If funding dries up – it’s impossible to efficiently search for novel antimicrobial agents; determine which antimicrobial agents are worth testing; and evaluate these antimicrobial agents in animals/humans.
Clinical trials: Designing large-scale clinical trials to evaluate new antibiotics in tuberculosis is relatively difficult. The trials need to recruit large numbers of people that are infected with TB and convince some of these individuals to agree to receive an experimental treatment.
Antibiotic resistance: Tuberculosis bacteria can develop antibiotic resistance via numerous mechanisms such as: mutation (during replication); gene transfer (microbes transfer genes to each other within the body); and selective pressure (microbes with resistance genes survive).
- As a result, even if we have a new highly-effective antibiotic for TB – it’s likely a matter of time before TB strains develop resistance to it.
- Irresponsible antibiotic use can accelerate antibiotic resistance. (This is a huge problem in developing countries due to the fact that many people simply self-prescribe antibiotics because they’re available OTC).
Not profitable: Developing new antibiotics is not profitable for pharmaceutical companies – hence the abandonment of new antibiotic development by most. Why isn’t it profitable? A variety of reasons…
- Cost: It requires hundreds of millions of dollars (possibly eclipsing $1B) to: research new prospective antibiotic compounds, test the compounds, put promising compounds through clinical trials, etc.
- Time: It takes many drugs 10-15 years to complete all 3 phases of clinical trials. (Many drugs fail at one stage of trials – such that the time/money investment was wasted.)
- Poor countries: Tuberculosis is an infection that disproportionately affects poorer, developing countries. The parts of the world that need new antibiotics most cannot afford to pay top dollar for the latest flashy antibiotic (even if it’s really good). So what happens?
- Negotiate a deal: Many countries will negotiate a favorable deal with the pharmaceutical companies that developed the new antibiotic such that it’s affordable but doesn’t come close to helping the pharmaceutical company recoup their total investment.
- Patent theft: Certain countries just don’t care about ripping off new antibiotic patents if they’re able to – and manufacture them on their own. After all, these drugs are needed to save lives – and many agree that saving lives is probably more important than patent theft.
- Short-term use: Most antibiotics are not used for a long-term in the treatment of bacterial infections. Sure tuberculosis is treated for longer than most bacterial infections (6-21 months depending on the case) – but short-term antibiotic use is not as profitable as long-term/chronic use (such that pharma R&D is better focusing on chronic diseases like type-2 diabetes, hypertension, high cholesterol, etc.).
- Wealthy countries: Tend to have low rates of tuberculosis. Most pharmaceutical companies make a majority of their money targeting U.S. consumers. The U.S. essentially subsidizes drugs for the rest of the world. If they can’t recoup any investment from the U.S. populace (where TB rates are low) it doesn’t incentivize anti-tubercular drug development.
Why am I interested in tuberculosis?
Because it’s directly related to a pathogen that once (somehow) infected me – despite having zero risk factors (immune function normal; bodyweight normal; lung function normal; no genetic risk factors; never smoker; etc.).
I was infected with Mycobacterium avium complex (MAC) – likely due to exposure via aerosolized water or soil – and had to take three high-dose antibiotics for ~18 months (azithromycin, rifampicin, ethambutol) in hopes of eradicating my lung infection.
Thankfully the antibiotics managed to “cure” my infection – but they were extremely difficult to tolerate for such a prolonged duration.
Although I’m still more interested in MAC than MTB for personal reasons – I was completely oblivious to the ongoing devastation/destruction tuberculosis continues to cause (at the time of my infection I didn’t even know MTB still existed – I thought humanity “cured” it long ago).
And although I’d like to travel to a variety of countries in the future, my selection may be limited given that my personal risk tolerance is low – and I don’t want to risk a TB infection by traveling to a place with high rates of TB (especially if they have high rates of drug-resistant TB.)
Do you have any ideas for new TB treatments?
- Where would you direct focus/effort for fastest ROI in TB research?
- Have you ever been treated for tuberculosis?
- Latent or active? (If active, how severe?)
- Drug-sensitive or drug-resistant?
- What antibiotic regimen was prescribed?
- Did you respond well to treatment?
- Did you experience treatment-related side effects?