There is a considerable amount of evidence suggesting that pepsin, a common digestive enzyme, is involved in the pathogenesis/pathophysiology of laryngopharyngeal reflux (LPR) disease. (R)
Specifically, it is thought that pepsin ends up refluxed into the laryngopharynx from the stomach whereafter it causes damage, inflammation, and dysfunction of the: cells, tissue, mucosal membranes, nerves, and/or upper esophageal sphincter (UES).
For this reason, researchers like Nikki Johnston et al. thought to investigate potent pepsin inhibitors fosamprenavir and darunavir (FDA-approved for the treatment of HIV-1 infection) – for the treatment of laryngopharyngeal reflux (LPR) disease.
Why investigate fosamprenavir for LPR? (Rationale)
There is some strong rationale for investigating the efficacy of fosamprenavir and other HIV protease inhibitors (amprenavir, ritonavir, saquinavir, darunavir) for the treatment of LPR.

Efficient & long-lasting pepsin inhibitor
Pepsin is understood to be implicated in the pathophysiology of LPR – such that it may cause LPR onset (to some extent) and/or may sustain LPR disease (to some extent).
Various substances are thought to inhibit pepsin such as sucralfate, alginate, and even alkaline water (high pH).
However, the efficacies of sucralfate, alginate, and alkaline water at providing: (1) significant (i.e. high magnitude) and (2) sustained (i.e. prolonged) – inhibition of pepsin are probably low.
Fosamprenavir effectively and efficiently inhibits pepsin – with IC50 in the low micromolar range, meaning not much of fosamprenavir is needed to block pepsin-mediated damage.
Fosamprenavir also provides a long-lasting effect after being metabolized into amprenavir (with an elimination half-life of ~7.7 hours).
The unique combination of pepsin inhibition potency AND prolonged duration of action are what make fosamprenavir an appealing investigational drug for LPR.
Low rates of LPR in HIV patients
According to Johnston et al., patients using HIV protease inhibitors have far lower rates of LPR (0.2%) than the general population (10-34.4%). (R)
This suggests that a major reason as to why LPR rates might be lower-than-average in HIV patients could be due to regular/chronic use of HIV protease inhibitors.
How could this be? Protease inhibitors like fosamprenavir should theoretically bind to and deactivate pepsin (and possibly other enzymes) such that even if refluxed into the laryngopharynx – no laryngeal damage occurs because the pepsin is inactive.
This may interfere with the pathogenesis and/or pathophysiology of laryngopharyngeal reflux disease (LPRD) such that it never develops or is significantly less likely to develop while using protease inhibitors.
Although low rates of LPR in HIV patients is an interesting observation, we must acknowledge that this is merely a correlation – and may be explainable by factors other than use of protease inhibitors like fosamprenavir.
It’s possible that laryngopharyngeal reflux events still occur in these patients but pepsin doesn’t do significant damage (due to ongoing protease inhibitor use) – such that discontinuation of protease inhibitors would lead to rapid onset LPRD in some patients.
It’s also possible that HIV patients exhibit lower body weight (for various reasons) and are thus less likely to have conditions like hiatal hernia and/or overweight/obesity-related reflux – which are common in many cases of LPR.
Efficacy in animal models of LPR
Johnston et al. (2022) published a study in the journal of Laryngoscope entitled “Oral & Inhaled Fosamprenavir Reverses Pepsin-Induced Damage in a LPR Mouse Model.”
In this study researchers created a mouse model of LPR (mechanical injury once weekly for 2 weeks and pH 7 solvent/pepsin instillation 3 days/week for 4 weeks).
Next they administered a protease inhibitor (fosamprenavir or darunavir) via gavage (tube leading down throat) or aerosol to the mouse model of LPR for a total of 4 weeks (5 days each week).
Results indicated that gavage and aerosol fosamprenavir prevented pepsin-induced laryngeal damage (reactive epithelia, intraepithelial inflammation, cell apoptosis, etc.).
Although this was an ultra-specific mouse model of LPR (may not translate well to human LPR) – this provided preliminary evidence necessary to justify investigating fosamprenavir in humans with LPR.
No effective LPR treatments
First-line recommendations for the treatment of LPR include: PPIs or H2 blockers; antacids; alginates; lifestyle changes (weight loss, exercise, head-of-bed elevation, looser clothing, stress reduction); and dietary adjustments (lower acid, smaller meals, no eating 4+ hours before bed, etc.).
Despite these recommendations, none are really supported by high-quality (i.e. strong) scientific evidence.
For this reason, trying to manage symptoms of LPR can turn into somewhat of a crapshoot – ongoing self-experimentation with various medications, supplements, dietary adjustments, and possibly even surgeries.
Considering that ~10% of the U.S. population (1/10 people) have laryngopharyngeal reflux (LPR) and no treatments have proven highly effective – there’s a significant need for innovation and development of new pharmacological therapies for this condition.
How fosamprenavir may treat LPR (mechanism of action)
Fosamprenavir is a prodrug that is hydrolyzed by cellular phosphatases to the antiretroviral protease inhibitor amprenavir.

The hydrolysis of fosamprenavir facilitates a slow release of the active compound amprenavir over an extended duration.
This is preferable to conventional amprenavir which facilitates only an acute or short-term effect (and thus requires several frequent doses to achieve the same effect as a single dose of fosamprenavir).
Amprenavir is understood to function as a protease inhibitor wherein it interferes with enzymes that cleave proteins.
HIV protease inhibitors including: amprenavir, ritonavir, saquinavir, and darunavir effectively bind to and inhibit pepsin with IC50 in the low micromolar range.
This means that even small concentrations of protease inhibitors like fosamprenavir will inhibit pepsin – which is relatively significant given the fact that there aren’t any well-known pepsin inhibitors with potencies necessary to appreciably deactivate pepsin.
The targeted administration of fosamprenavir (which gets converted into amprenavir after ingested) to the laryngopharynx via inhalable suspension – may reverse or attenuate pepsin-mediated laryngopharyngeal damage.
The reversal or attenuation of pepsin-mediated laryngopharyngeal damage by fosamprenavir (or other protease inhibitors) may significantly improve LPR symptoms and, in select cases, could entirely reverse laryngopharyngeal reflux (LPR) disease.
Fosamprenavir for LPR (Research)
Thus far I was able to find just one study in which the effect of fosamprenavir was evaluated for the treatment of LPR in mouse models (with mechanically-induced laryngeal injury and subsequent pepsin instillation).

Oral & Inhaled Fosamprenavir Reverses Pepsin-Induced Damage in a LPR Mouse Model
Johnston et al. (2022) (R)
Aim: Identify small molecule inhibitors of pepsin and test their efficacies to prevent pepsin-mediated laryngeal damage in vivo.
Method: Evaluate drugs for pepsin binding and inhibition via high-throughput assays and crystallography; create a mouse model of LPR (involving pepsin); administer pepsin inhibitors identified and determine efficacy.
Results: HIV protease inhibitors (amprenavir, ritonavir, saquinavir, darunavir) bind to and inhibit pepsin with IC50 in the low micromolar range. Gavage and aerosolized fosamprenavir blocked pepsin-induced laryngeal damage (e.g. reactive epithelia, intraepithelial inflammation, cell apoptosis). Darunavir gavage exhibited mild reactivity without significant protection whereas aerosolized darunavir prevented cellular apoptosis.
Conclusion: Fosamprenavir and darunavir, FDA-approved meds for HIV/AIDS bind to and inhibit pepsin – offsetting pepsin-induced laryngeal damage in a LPR mouse model. Reformulation of fosamprenavir for local inhaled delivery might improve outcomes and minimize side effects.
N-Zyme Biomedical (statement)
N-Zyme has confirmed efficacy of fosamprenavir in an in vivo model.
Co-crystallization studies, competitive binding assays, and an enzymatic assay revealed select HIV protease inhibitors bind to the active site of pepsin and inhibit the enzyme.
Fosamprenavir was deemed the lead candidate and subsequently tested in an in vivo model of LPR to assess its efficacy to abrogate pepsin-mediated laryngeal inflammation and damage.
Oral administration of fosamprenavir (20 mg/kg/day) – equivalent to the FDA-approved dose used to treat HIV in humans, prevented pepsin-mediated laryngeal damage.
Administration by inhalation at a lower dose of 1 mg/kg/day also prevented pepsin-mediated laryngeal injury.
In support, using the Cohort Discovery Tool of the Medical College of Wisconsin Clinical Data Warehouse, we found patients taking HIV inhibitors have significantly lower incidence of LPR (0.2%) compared to that reported for the general population (10-34.4%).
Limitations of this study
The primary limitation of this study is that fosamprenavir was evaluated in mice (not humans) with a highly specific LPR model.
It’s possible that fosamprenavir is somehow more potent and/or effective in mice in deactivating laryngeal pepsin than in humans – even though I think this is unlikely.
Additionally, even if pepsin does play a role in causing and/or sustaining a subset of LPR disease cases – it’s possible that pepsin deactivation with fosamprenavir in humans with pepsin-implicated LPR will not derive clinically significant benefit from its administration.
Lastly, it should be obvious that subtypes of LPR wherein pepsin is NOT causally implicated (or not implicated to a significant extent) – fosamprenavir will probably be relatively useless.
N-Zyme Biomedical: Repurposing Fosamprenavir for LPR
N-Zyme Biomedical is a pharmaceutical company spearheaded by pepsin researcher Nikki Johnston (PhD) with the likes of Jamie Koufman (ENT who coined the term “LPR”) on the advisory board.
2 formats of fosamprenavir for LPR:
Oral fosamprenavir: N-Zyme partnered with LGM Pharma to develop an oral version of fosamprenavir containing alginate to prolong its delivery. This will provide a systemic effect (and thus probably cause more side effects).
Inhaled fosamprenavir: N-Zyme developed a dry powder form of fosamprenavir for administration via a specialized inhaler. This contains a low-dose of fosamprenavir and aims to deliver a highly-localized action (such that there’s no appreciable systemic effect).
It makes logical sense that N-Zyme Biomedical is testing 2 distinct formats of fosamprenavir.
- One format may work well for LPR and the other may prove to be completely ineffective… by testing both at least you don’t miss out on an effect from either the oral or inhalable format.
- One format may prove significantly more effective for LPR than the other – such that by testing both at least you won’t miss out on a version that was significantly more effective than the one that gets approved.
- The oral version of fosamprenavir is slightly tweaked and contains alginate (which also inhibits pepsin to some extent and prolongs the duration of action for fosamprenavir).
- I view alginate as a major bet hedge considering that alginate alone often provides some efficacy in LPR.
- I dislike the combining of alginate with oral fosamprenavir because it makes it more difficult to determine the standalone efficacy of fosamprenavir vs. oral alginate (such that alginate may be providing most or all of the therapeutic benefit).
- I would like to see concentrated alginate (as a control) compared to oral fosamprenavir in a double-blind, randomized, controlled trial for LPR.
- Inhaled fosamprenavir could prove to be extremely lucrative given the fact that they’ll be selling a specialized medical device along with the new powdered fosamprenavir.
According to N-Zyme Biomedical:
- LPR is estimated to affect more than 20% of the U.S. population.
- $26B/year spent on PPIs for LPR despite no RCTs showing efficacy.
- No medical therapy is available for LPR.
Currently N-Zyme has: (1) Internal patent application filed; (2) FDA/IND Phase 3 clinical trial approval; and (3) IRB Phase 3 clinical trial approval from the Medical College of Wisconsin.
In other words, N-Zyme Biomedical has been given the “green light” for Phase 3 trials to assess the efficacy of fosamprenavir for the treatment of LPR.
N-Zyme Biomedical states (R):
Or cutting-edge research demonstrates fosamprenavir binds to and inhibits pepsin, preventing pepsin-mediated inflammation and tissue damage.
Using an innovative delivery approach, N-Zyme Biomedical aims to provide local inhaled delivery of a low dose of fosamprenavir directly to the throat providing the first safe, effective, and efficient treatment for the millions of people that suffer from LPR, addressing a significant unmet clinical need.
As of May 2022, N-Zyme Biomedical announced a partnership with LGM Pharma to “create a new, superior formulation of oral/systemic fosamprenavir, unique to N-Zyme, for the treatment of LPR.”
The new formula will contain alginate to prolong drug delivery. They hypothesize that this will improve its efficacy for reflux.
As a result of this formula adjustment, this has delayed the start of Phase 3 clinical trials until early 2023 – but they believe it will allow them to submit a new drug application (NDA) to the FDA for the systemic medication at the conclusion of Phase 3 while they simultaneously perform clinical trials for a dry powder inhaler to provide local delivery of a low dose.
The planned end date for the trial will be sometime in 2024.
Leadership
Franco Vigile (Co-Founder & CEO): Entrepreneur with over 10 years experience in founding/operating successful companies.
Nikki Johnston (CSO): 20 years experience in LPR and pepsin research, supporting the clinical trial of repurposed fosamprenavir for LPR. Part of the team preparing international evidence-based guidelines for the definition and diagnosis of LPR.
Advisory board
- Jamie Koufman (MD): Nearly 4 decades of clinical and scientific research on the diagnosis, treatment, and biology of reflux conditions. Credited with coining the terms “laryngopharyngeal reflux,” “silent reflux,” “airway reflux,” and “respiratory reflux.”
- Prakash Gowd (B.Sc. Pharm): Executive management and strategic business analysis in healthcare and biopharma.
- Thomas L. Carroll (MD): Researcher of glottic insufficiency and LPRD. Special interest in non-acid reflux and chronic cough.
- Jerome R. Lechien (MD, PhD, M.Sc.): Otolaryngologist and professor of otolaryngology.
- Jayne E. Hastedt (PhD): Experience in pharmaceutical product development.
- Brian Jahns (B.Sc., PharmD): Medical marketing and sales leadership.
- Dana Nohynek (M.Sc., RAC): Developer of regulatory strategies during clinical trials.
Clinical trial of Fosamprenavir (Lexiva) for LPR (Details)
Included below are details about the upcoming clinical trial of fosamprenavir that will start sometime in 2023 and will be completed probably sometime in 2024-2025. (R)
Aim: Evaluate the efficacy of fosamprenavir/Lexiva for LPR in a 12-week, randomized, double-blind, placebo-controlled trial.
Methods: Compare fosamprenavir (Lexiva) [at the FDA approved, manufacturer-recommended dose for HIV] for 12 weeks in medically-refractory patients with clinically diagnosed moderate/severe LPR vs. a placebo control.
Sample: 104 LPR patients (52 will receive fosamprenavir & 52 will receive a placebo).
Outcome measures: RSI (Reflux Symptom Index). RFS (Reflux Finding Score). Multi-channel intraluminal impedance pH (MII-pH). Saliva (pre & post-treatment) to analyze pepsin levels.
Potential benefits of fosamprenavir for LPR

May effectively manage LPR
- Fosamprenavir may not necessarily “cure” LPR in all cases due to the fact that some people have mechanical issues like large hiatal hernia that generate substantial regular reflux.
- As a result of these mechanical issues, stomach contents like: trypsin, pepsin, hydrochloric acid, bile salts, etc. will keep getting refluxed into the laryngopharynx such that pepsin continues to accumulate in the laryngopharynx.
- That said, the ongoing administration of fosamprenavir in patients with LPR who are unable to correct mechanical, neurological, and/or motility issues with surgery – may continue neutralizing the damaging effect of pepsin such as to significantly reduce symptoms and improve quality of life.
May cure LPR
- It is thought that a subset of individuals with LPR will never fully reverse the condition because they’re unable to fully deactivate and/or eradicate pepsin from laryngopharyngeal tissue.
- Interventions like sucralfate, alkaline water, alginates, chewing gum, etc. do not have the same potency and/or duration of action as fosamprenavir – such that they probably never fully eliminate intracellular laryngeal pepsin.
- As a result, each exposure to low or moderate pH foods/drinks continues to “reactivate” pepsin present in laryngopharyngeal tissues – causing cellular inflammation, apoptosis, etc. and the perpetuation of LPR disease.
- Assuming no mechanical and/or motility issues that caused the reflux – eradicating laryngopharyngeal pepsin with fosamprenavir could theoretically “cure” a percentage of LPR cases.
Cancer prevention
- Research suggests that pepsin may be a causative factor in the development of esophageal and/or laryngeal cancers occurring secondary to reflux conditions (e.g. GERD & LPR). (R)
- For this reason, deactivating pepsin in areas outside of the stomach (e.g. the esophagus) could potentially be useful as a cancer prophylactic in patients with GERD and/or LPR that are considered high risk (e.g. Barrett’s esophagus).
Few side effects (?)
- It’s possible that the aerosolized (i.e. inhaled) fosamprenavir will have few overall side effects for the treatment of LPR.
- Inhaled fosamprenavir should theoretically exert most of its effect locally (within the nasal cavities, pharynx, and larynx) such that systemic effect and subsequent side effects may be minimized or avoided.
- With the inhalable version of fosamprenavir I’d still expect some throat irritation and/or discomfort though – but it may be nothing compared to full-blown LPR symptoms.
Potential drawbacks of fosamprenavir for LPR
Cost
- If approved fosamprenavir will likely prove to be extremely expensive in the “novel” formats created by N-Zyme Biomedical et al.
- (One version uses a specialty “oral formula” and the other uses a specialty “powdered formula” with a patented delivery device.)
- Considering that fosamprenavir oral medication costs ~$350-800 most places – I’d expect the LPR version to be even more expensive.
- If approved, it is likely that N-Zyme will make some serious bank from those with good insurance and/or “coupons” (enabling tax cuts).
Interactions
- Fosamprenavir is metabolized into amprenavir by CYP3A4 – and such it is a modest CYP3A4 inhibitor.
- Any co-administered substances subject to CYP3A4 metabolism could cause pharmacokinetic interactions.
Ineffective (certain LPR cases)
- Despite the fact that many cases of LPR exhibit abnormally high levels of laryngopharyngeal pepsin – not all LPR cases present with pepsin.
- The subset of LPR cases wherein pepsin is not appreciably implicated won’t respond to fosamprenavir (as it’s entire purpose is to deactivate pepsin).
Side effects
- Common side effects include: diarrhea; nausea; upset stomach; headache; and fatigue.
- Long-term effects can include: hyperlipidemia and lipodystrophy. (R)
- There are also potentially unknown side effects that may result from an aerosolized version of fosamprenavir (despite using a lower dose and having a more localized distribution).
How pepsin may cause & sustain LPR disease (details)
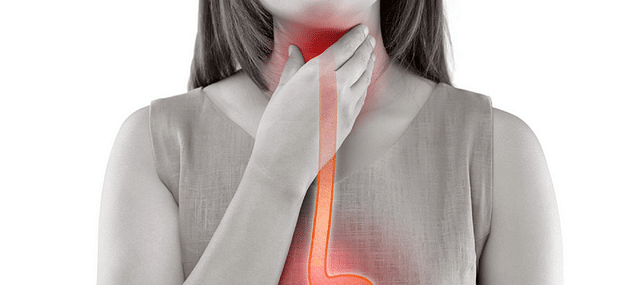
1. Pepsin gets refluxed into the laryngopharynx
Although it’s unlikely that pepsin is the only cause of LPR – it may be a highly-significant causative variable.
With LPR, it’s likely that something happens to induce increased reflux (e.g. hiatal hernia) and/or reflux is particularly acidic (such as due to regularly consuming acidic foods/drinks).
As a result, a substantial amount of pepsin gets refluxed into the laryngopharynx and/or the nasopharynx and lingers in tissues within this region.
2. Pepsin induces damage & inflammation
Pepsin damages tissues and causes cellular death within the throat, nose, and esophagus – particularly in the presence of other refluxate (e.g. hydrochloric acid).
Kowalik & Krzeski: “Pepsin initiates inflammatory changes within the larynx, nasopharynx, and nasal cavity.” (R)
This can actually cause seriously uncomfortable symptoms like: ear pain; burning behind the eyes; and dizziness or disequilibrium.
It also seems to change gene expression and decrease expression of protective proteins within laryngopharyngeal tissue – such as to increase susceptibility to further damage.
Chronic pepsin-mediated damage may also cause esophageal motility disorders, damage laryngeal nerves, and/or cause cancer onset – all of which perpetuate LPR disease.
Essentially a damaged or inflamed laryngopharynx does not function normally and this causes symptoms like: globus sensation (i.e. feeling a lump in throat), swallowing difficulty, and prolonged motility.
3. Pepsin stays lodged in the laryngopharynx
A major problem with pepsin entering the laryngopharynx via reflux events is that laryngopharyngeal tissue did not evolve to accommodate activated pepsin like the stomach – and predictably it becomes damaged.
Pepsin also remains stored intracellularly for up to 24 hours, is stable up to pH 7, and rapidly regains activity after reacidification.
This is why some individuals who undergo surgery for LPR still exhibit high concentrations of pepsin in the laryngopharynx post-surgery (even if the surgery corrected the underlying cause of reflux).
In other words, even if zero additional pepsin gets refluxed into the laryngopharynx – the old pepsin remains because it continues to “reactivate” with exposure to even moderate pH foods/drinks (below 7 pH).
4. LPR symptoms continue
Since pepsin lodged in the laryngopharynx keeps “reactivating” each time a person consumes foods and/or drinks with a pH below 7 – LPR symptoms may continue indefinitely unless the pepsin is eradicated.
Even if a person strictly follows a “low acid diet” wherein all foods and drinks are alkaline (i.e. non-acidic) – this may help in terms of reducing pepsin activation but it won’t necessarily eradicate the pepsin.
The logic behind a low acid diet (i.e. alkaline diet) is that low acid foods/drinks won’t activate pepsin on the way down the esophagus to trigger symptoms (whereas acidic foods/drinks will instantly reactivate laryngeal pepsin if swallowed – and cause symptom flares).
5. Only partial or incomplete recovery
For this reason, people who: (1) take PPIs to decrease the acidity of reflux events and (2) consume low acid diets (i.e. alkaline foods/drinks) may report some degree of LPR improvement – but not full LPR remission.
The partial or incomplete recovery might be explained by the continued presence and reactivation of pepsin in laryngopharyngeal tissues.
Once present (or after a certain threshold of pepsin is present) it seems nearly impossible to completely eradicate in some individuals.
LPR disease gets even worse if reflux events are frequent and/or highly acidic due to the fact that the refluxate composition may contain significant pepsin – which adds fuel to the fire (more pepsin to the already-present pepsin).
Pepsin as a biomarker for LPR diagnosis…
Pepsin is considered to be a sensitive and specific biomarker for LPR. (R)

This is why a rapid, non-invasive diagnostic test called “Peptest” was developed to detect the presence of pepsin and confirm clinical suspicion of LPR.
If pepsin is detected in the laryngopharynx and LPR symptoms/signs are present (based on subjective reports & objective laryngeal examination), LPR diagnosis is likely accurate.
The larynx has a pH of ~6.8 and pepsin remains active at a pH up to ~7 – meaning it will remain active in the larynx unless deactivated (such as with a pepsin inhibitor like fosamprenavir).
Pepsin in isolation probably doesn’t do significant damage to the laryngopharynx, however, pepsin combined with hydrochloric acid, bile salts, and/or other refluxate will “re-activate” pepsin and perpetuate laryngopharyngeal symptoms.
Considering the fact that pepsin may play a significant role in causing and sustaining LPR symptoms – targeting pepsin with a pepsin inhibitor may prove therapeutic for the treatment of LPR.
Fosamprenavir for LPR (Reactions)
Included below are reactions from individuals who have LPR and reacting to the mention of fosamprenavir being potentially useful int eh treatment of LPR.
Reaction #1
I started taking PreP because I’d hoped it would help with LPR too (and well also because I’m gay) but it hasn’t made a noticeable difference. Maybe it doesn’t have the same pepsin-mediating properties of other HIV drugs?
My response…
PrEP (i.e. pre-exposure prophylaxis) contains a fixed-dose combination of emtricitabine/tenofovir. Neither of these compounds are protease inhibitors.
- Emtricitabine functions as a reverse transcriptase inhibitor (the enzyme that copies HIV RNA into new viral DNA).
- Tenofovir functions as a nucleotide analogue reverse-transcriptase inhibitor (NtRTI) by selectively inhibiting viral reverse transcriptase.
Even if these were protease inhibitors – it doesn’t mean they’d necessarily be effective for the deactivation of pepsin.
In an animal model study involving pepsin-mediated LPR, only fosamprenavir significantly reduced pepsin-mediated damage – indicating potential differences in ability to neutralize pepsin among protease inhibitors.
Reaction #2
If I am reading the clinical trial information correctly, they are repurposing an HIV protease inhibitor prodrug called fosamprenavir (active form, amprenavir).
Old data suggested chemical/structural similarities to drugs that inhibit aspartyl proteases like pepsin and some research has confirmed it.
But it isn’t a strong effect and may require high doses due to an apparently high Kd (low affinity).
However, it does get into cells which might offer some advantage over alkaline solutions (since cells buffer their pH pretty well) – so maybe it could help in that capacity as there is some evidence that pepsin gains entry into cells in the throat and nose as opposed to simply resting on the surface of these tissues.
However, the drug that will most likely be used is actually Darunavir.
Dr. Johnston has identified 4 different protease inhibitors that can inhibit pepsin, and Darunavir has the lowest (best) IC50 for pepsin of the 4 meaning only 0.06 uM of the drug is needed to inhibit half of any pepsin present.
These protease inhibitors are ideal for repurposing because they target a foreign virus so they will have much fewer side effects compared to repurposing other drugs.
Local delivery by inhaled aerosol will also allow for a reduced dose of the drug (as opposed to the high doses needed to get enough of the drug from the GI tract via bloodstream to laryngeal tissue) – and may prevent side effects since it won’t go through the GI tract.
Reaction #3
What the heck man, 2 years to do this study?! I’m gonna have to somehow fake having HIV to get this drug.
Will fosamprenavir work for all cases of LPR?
Unlikely. Patients without pepsin-positive LPR are unlikely to derive any significant benefit from fosamprenavir.
Some will question whether patients who don’t exhibit pepsin in the larynx, pharynx, and/or nasal cavities legitimately have LPR – and this is a valid question.
If detectable pepsin is a requirement for valid LPR diagnosis – then fosamprenavir has potential to provide symptom relief for all patients with LPR.
However, at this time it is unclear as to how effective fosamprenavir will be in humans at deactivating pepsin in LPR – and whether the deactivation of pepsin actually reduces LPR symptoms.
Will fosamprenavir cure LPR disease?
Maybe a subset of cases – such as patients with: (1) detectable pepsin and (2) pepsin causally implicated in the pathogenesis and/or pathophysiology of LPR disease.
Fosamprenavir may cure select cases of LPR by eradicating the presence of pepsinogens in the laryngopharynx.
The eradication of pepsinogens may, for select individuals, enable healing of damaged cells/tissue and restoration of normative UES function – such that no more reflux occurs.
In patients with an underlying health issue that’s causing LPR as a secondary effect such as: hiatal hernia; esophageal motility disorders; eosinophilic esophagitis; laryngopharyngeal infection; functional esophageal disorders; etc. – fosamprenavir will not “cure” the condition.
Why? Because these patients will continue “refluxing” up gastric contents (e.g. gastric acid, bile salts, trypsin enzymes, pepsin enzymes, undigested food particles, etc.) such as to sustain the pathophysiology (i.e. diseased state) of LPR.
Read: Can Reflux Be Cured?
Even if pepsin is the sole cause of one’s LPR symptoms (e.g. hoarseness, coughing, globus sensation, etc.) this does NOT mean using fosamprenavir to eradicate or manage symptoms is a complete cure.
For example: A large hiatal hernia may cause constant reflux throughout the day.
Even if fosamprenavir effectively attenuates pepsin-mediated symptoms – one might need to use fosamprenavir for life unless the root causes of reflux are corrected (e.g. hiatal hernia surgery).
That said, it’s possible that regular use of inhalable fosamprenavir will significantly improve symptom severity and quality of life for many patients with LPR – even if they never are able to fix the underlying cause(s) like hiatal hernia.
Any other pepsin inhibitors to try for LPR?
With regard to pepsin inhibition in LPR, we must consider the: (1) magnitude of inhibition (IC50 value) and (2) duration of inhibitory effect.

Most substances either don’t provide adequate pepsin inhibition and/or exhibit a short-term inhibitory effect – such that pepsin continues to reactivate.
Additionally, even if using an inhibitor, it’s possible to potentially overpower the inhibitory effect such as by consuming acidic foods/drinks, etc.
Note: The links below are affiliate links – the cost is the same regardless and these help support the site.
Alkaline water
I recommend Essentia simply because it seems to be the best value for the money on Amazon.
This is the brand that I personally use for my LPR – but other brands are probably fine too.
Some people have gone as far as to buy their own alkaline water maker for the sake of saving money – but if you go this route, make sure that it’s actually making alkaline water (test it).
Baking soda
Mark Noar (GI surgeon & LPR specialist) recommends mixing a combination of alkaline water and baking soda to create a highly alkaline solution.
This alkaline solution is then: gargled (in the back of the throat), sprayed up the nostrils, and/or sprayed in the back of the throat – you can mix and put into your own nasal spray bottles just be sure to avoid bacterial contamination.
Can also consider experimenting with Arm & Hammer Nasal Spray after meals – as this stuff contains sodium bicarbonate in the solution.
Unlike just using alkaline water which quickly rinses off of tissues – baking soda helps “coat” the nasal cavities, larynx, and pharynx – to neutralize pepsin for a longer duration.
Some people drink baking soda and claim that it helps with reflux – but those consuming baking soda should beware of the maximum safe threshold per day (and avoid exceeding this amount).
Curcumin
There’s some evidence that curcumin (10 micromol/L) inhibits peptic activity and pepsin-induced mitochondrial damage.
The problem with curcumin though is that most formulations contain “black pepper extract” (i.e. bioperine) which will reactivate pepsin and worsen symptoms.
A person would need to essentially administer curcumin intracellularly specifically to laryngeal tissue.
Curcumin is not very bioavailable such that oral supplementation is unlikely to significantly affect pepsin in laryngopharyngeal tissue.
It’s possible that one could create a curcumin solution mixed with alkaline water (analogous to baking soda/alkaline water mix) but I’m unsure as to how efficiently curcumin penetrates laryngeal tissue to neutralize intracellular pepsin.
Nikki Johnston was involved in research suggesting that phytochemicals (one of which was curcumin) may be useful for offsetting pepsin-mediated damage in LPR. (R)
Sucralfate
Individuals struggling with pepsin-confirmed LPR should consider trying sucralfate.
Sucralfate is a medication that’s commonly used to treat stomach ulcers, GERD, and stomach inflammation.
It’s a locally acting drug that reacts with hydrochloric acid in the stomach to form a viscous, paste-like material that functions as an acid buffer within 1-2 hours of administration and for up to 6-8 hours post-administration.
Alginates (Gaviscon Advance & Reflux Gourmet)
Alginate (alginic acid) is a naturally-occurring edible polysaccharide found in brown algae.
Preliminary evidence and a significant number of anecdotes suggest that when administered after meals – it forms a barrier against reflux-related damage.
Evidence suggests that alginate formulations (e.g. Gaviscon Advance) coats the esophagus and neutralizes pepsin and forms a barrier in the esophagus to prevent reflux events.
I’ve tried both Gaviscon Advance and Reflux Gourmet – the former is more effective and contains more alginates and the latter is less effective but tastes way better.
Magaldrate
Magaldrate is a combination of magnesium carbonate and aluminum hydroxide – both of which rapidly neutralize stomach acid after meals.
Although this is an antacid that is recommended to be used post-eating – evidence suggests that it may deactivate and adsorb pepsin.
Magaldrate also binds a considerable amount of substances in reflux conditions such as bile acids and lysolecithin.
Zinc L-Carnosine
Contains 23% zinc and 77% L-carnosine by mass and is an approved drug in Japan for the treatment of gastric ulcers.
Research suggests that zinc L-carnosine stimulates mucus production and maintains the integrity of the gastric barrier.
Some claim that it exerts a similar effect to sucralfate – and if true – it may help deactivate and/or neutralize pepsin.
However, because this supplement is not sold in a liquid suspension format to coat the laryngopharynx – it may not be very effective at counteracting laryngeal pepsin.
Chewing gum
There’s some evidence that chewing gum may help LPR – I personally chew unflavored gum and sometimes juicy fruit.
How? Chewing gum increases saliva production and esophageal motility – both of which can increase pH and neutralize acidity.
By decreasing overall acidity in the laryngopharynx via gum chewing, pepsin should theoretically be less active/deactivated.
Just make sure to avoid chewing gum with acidic components or with components that cause LES relaxation (e.g. mint).
Other pepsin inhibitors
- Ecabet sodium: Ecabet sodium (125 microg/mL) seems to inhibit pepsin. These also seem to be useful in the treatment of functional dyspepsia (a common functional esophageal disorder). (R)
- Black raspberry extract: Anthocyanin-enriched black-raspberry extract (100 mcg/mL) appears to effectively inhibit pepsin.
- Marial: This is a drug approved in Europe for the treatment of GERD & LPR. It contains hyaluronic acid, hydrolyzed keratin mucoproteins, glycoproteins, meso glycans and integrins, and viscous agents. (It seems to repair & regenerate mucous membranes in the esophagus via adhesive film-forming properties – and may inhibit pepsin). (R)
How I’d use fosamprenavir for LPR
If I were dealing with a case of LPR, below is how I’d personally test fosamprenavir as a treatment.
1. Confirm presence of pepsin in LPR
Before using fosamprenavir, I’d probably want to consider testing for the presence of pepsin by using something like Peptest.
Why? Because if pepsin is not detectable – I may want to reconsider the accuracy of LPR diagnosis and would probably avoid fosamprenavir (as it primarily will provide benefit by deactivating pepsin).
Because there’s no “official” diagnostic criteria for LPR – it’s possible that specific subtypes of LPR may exhibit no appreciable levels of pepsin in the larynx, pharynx, and nasal cavities.
Note: That said, just because pepsin isn’t detectable doesn’t automatically mean that no pepsin is present – tests may not be 100% reliable or able to detect all pepsin (e.g. intracellular pepsin).
2. Fosamprenavir trial
If pepsin is detected in airway specimens and/or saliva – I’d consider a trial of oral fosamprenavir with tempered expectations.
Why tempered expectations? Because oral fosamprenavir will be systemically distributed and thus only the amount of fosamprenavir that reaches the larynx, pharynx, and nasal cavities will provide an effect.
In theory fosamprenavir could be effective despite its systemic distribution based on the fact that it inhibits pepsin at very low (micromolar) concentrations.
I probably wouldn’t attempt grind the pills into powder and snort or inhale – as this could create unforeseen immune reactions, esophageal complications, etc.
I also probably wouldn’t grind up the fosamprenavir pills into powder and mix with alkaline water just to determine whether they’d effectively coat the larynx, pharynx, and nasal cavities.
Final thoughts on fosamprenavir for LPR
Fosamprenavir may be an effective treatment for a subset of patients with laryngopharyngeal reflux (LPR) in which pepsin is confirmed to present in the laryngopharynx.
At this time, it is unclear as to whether fosamprenavir will prove effective as a treatment for LPR in humans given that it was only tested on an ultra-specific mouse model of LPR.
Oral fosamprenavir pills (with systemic action) might not be effective relative to a strategically-formatted inhalable fosamprenavir (targeted action) that concentrates within laryngeal tissues to deactivate pepsin.
An inhalable version of fosamprenavir will likely enable high dose usage (relative to oral formats) while causing fewer side effects (due to localized action instead of systemic).
If I were eligible to get fosamprenavir, I’d probably evaluate its side effect profile and if it seems relatively low risk, I’d consider a 1-month trial.
If really desperate one could travel to a foreign country and likely have little difficulty buying fosamprenavir, however, it may be better to seriously consider surgery if that desperate.
If it works, then perhaps I’d tweak the dosage to find the lowest effective dosage necessary to effectively manage LPR symptoms – and then continue using it provided no long-term safety risks/concerns.
I’d then await an inhalable format (such as the one being tested by N-Zyme Biomedical) despite the fact that this will probably be very expensive.
The good news is that even if fosamprenavir proves to be completely ineffective for the treatment of LPR – it should theoretically inform us of the degree to which pepsin is responsible for LPR: (1) symptoms and (2) permanence/sustainment.
If fosamprenavir proves to be completely useless for LPR – we’ll be able to conclude that although pepsin is highly correlated with LPR, it’s more of a byproduct of LPR disease than an underlying cause.
Have you tried fosamprenavir for LPR?
Many people with LPR are desperate for some sort of symptom relief and would probably try anything (including off-label fosamprenavir) to treat LPR symptoms.
Keep in mind that systemically-administered fosamprenavir means it may not reach high enough concentrations within laryngopharyngeal tissue specifically to deactivate pepsin deposits in this region.
Thus, the only current format of fosamprenavir (oral) should not necessarily be expected to reduce symptoms of LPR.
If you have LPR and have tried fosamprenavir to treat symptoms – feel free to share some details in the comments.
- What format of fosamprenavir did you use for LPR? (Oral vs. inhalable)
- Did you find fosamprenavir to be effective for LPR treatment? (Rate efficacy on a scale of 1 to 10 with larger numbers = higher efficacy).
- How frequently did you administer fosamprenavir?
- Did you take a pepsin test (e.g. PepTest) to confirm the presence of pepsin?
- Would you participate in a clinical trial involving fosamprenavir for LPR?
Love the article. Long time LPR sufferer, even after fundo+HH surgery. Been stalking Fosamprenavir posts hoping for positive feedback.
One option would be to crush up the pills and mix a small dose with gaviston advance for more of a “coating”/local effect. But a systemic trial at 1400mg/day seems worthwhile. I just don’t know how I’d get a doctor in the USA to approve that.
Have you come across anyone who has tried it?